
Please note that the FDA lists medical device recall notices by the date that it posts the recall rather than the recall initiation date. If you wish to find information on a recall, or a correction or removal action that has not yet been classified, you can search the Medical Device Recalls Database. The links give details about what to do if you own or use one of these products. Use the yearly lists to find information about Class I medical device recalls and some Class II and III recalls of interest to patients. These products are on the list because there is a reasonable chance that they could cause serious health problems or death. According to one study, 28% of patients require pacemaker about 5 days after transcatheter aortic valve replacement.The FDA posts summaries of information about the most serious medical device recalls. One beneficial application for leadless devices may be postoperatively following transcatheter aortic valve replacement.

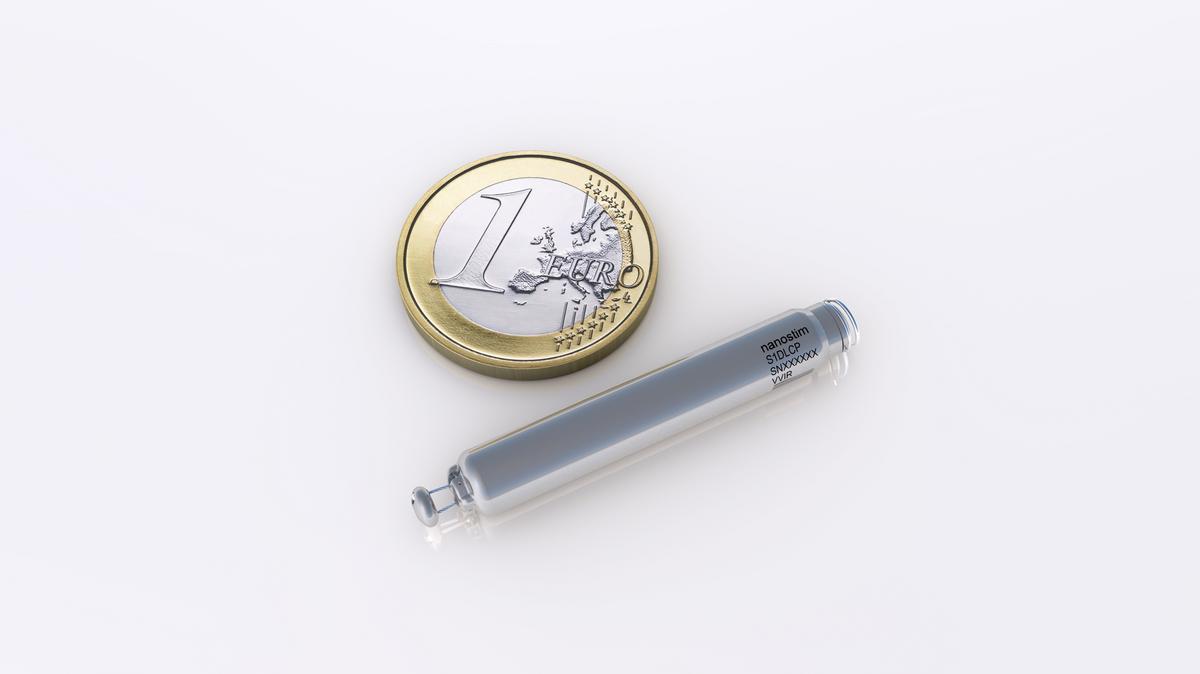
Current leadless pacemakers are designed to be compatible with magnetic resonance imaging. The recall described in this notice is the same one that was announced in the Stop New Implants of the Medtronic HVAD System Letter to Health Care Providers on June 3, 2021.Complications may occur related to femoral vein access or need for device repositioning there is moderate risk of cardiac perforation with subsequent pericardial effusion.Low-molecular weight heparin is administered preoperatively and during the procedure to prevent development of thrombosis.Nanostim attaches via an active screw-in helix and secondarily via three nitinol tines angled perpendicularly to the helix. Micra attaches to the right ventricle myocardium via four linear self-expanding nitinol tines.Only one leadless pacemaker (Micra ) has been approved by the US Food and Drug Administration for use in the United States a second (Nanostim ) is pending approval.
#MEDTRONIC PASSMAKER BATTERY RECALL UPDATE#
Saint Paul MN 55112: For Additional Information Contact: 76 Manufacturer Reason for Recall: Medtronic is informing doctors of two specific battery issues with EnRhythm pacemakers that will be addressed by a Medtronic software update available mid-2010. Leadless pacemakers are inappropriate for patients who require dual-chamber pacing, such as patients with certain forms of heart block or sinus node dysfunction. Cardiac Rhythm Disease Managment 8200 Coral Sea St. Leadless pacemakers may be suitable for patients with permanent atrial fibrillation with bradycardia or bradycardia-tachycardia syndrome or those who infrequently require pacing.
#MEDTRONIC PASSMAKER BATTERY RECALL GENERATOR#
The device is implanted via a femoral vein transcatheter approach it requires no chest incision or subcutaneous generator pocket. The leadless pacemaker, which is 90% smaller than a transvenous pacemaker, is a self-contained generator and electrode system implanted directly into the right ventricle.The following are key points to remember about this review of leadless pacemakers:
